
125
Learning Objectives
After studying this unit, students will be able to
Explain the properties of alkali metals and alkaline
earth metals
Recognise the anomalous properties of Li and Be
List the uses of alkali metals and alkaline earth
metals
Describe the general characteristics of compounds
of alkali metals and alkaline earth metals
Appreciate the biological importance of sodium
and potassium, Magnesium and Calcium
Explain the preparation, properties and uses of
calcium oxide, calcium hydroxide, gypsum and
plaster of paris.
Alkali and Alkaline Earth Metals
Unit
5
Rock salt
Sylvite
Spodumene

126
5.1 s-Block Elements:
The elements belonging to the group 1 and 2 in the modern periodic table are called
s-block elements. The elements belonging to these two groups are commonly known as
alkali and alkaline earth metals respectively. In this unit, we study their properties, uses,
important compounds and biological importance.
5.2 Alkali metals:
The word “alkali” is derived from the word al-qalīy meaning the plant ashes,
referring to the original source of alkaline substances. A water-extract of burnt plant
ashes, called potash contain mainly potassium carbonate. Alkali metal group consists
of the elements: lithium, sodium, potassium, rubidium, caesium and francium. They are
all metals, generally soft and highly reactive. They form oxides and hydroxides and these
compounds are basic in nature.
5.2.1 General characteristics of alkali metals:
Alkali metals are highly reactive and are found in nature only as compounds.
Rubidium and caesium are found associated in minute quantities with minerals of other
alkali metals. Francium is radioactive and does not occur appreciably in nature. Francium
is highly radioactive; its longest-lived isotope has a half-life of only 21 minutes.
Table 5.1 Abundance of important alkali metals and their sources
Elements
Abundance in
earth crust (%)
Relative
Abundance
Mineral source
Lithium 0.0018 35 Spodumene [LiAl(SiO
3
)]
Sodium 2.27 7 Rock Salt [NaCl]
Potassium 1.84 8 Sylvite [KCl]
Rubidium 0.0078 23
No convenient Source (obtained as
by product of lithium processing)
Cesium 0.00026 46
Figure 5.1 Alkali metals Li, Na and K stored under oil

127
Electronic configuration
The general valence shell electronic configuration of alkali metals is ns
1
, where ‘n’
represents the period number.
Table 5.2 Electronic configuration of alkali metals
Element Symbol Atomic No. Electronic conguration
Lithium Li 3 [He]2s
1
Sodium Na 11 [Ne]3s
1
Potassium K 19 [Ar]4s
1
Rubidium Rb 37 [Kr]5s
1
Caesium Cs 55 [Xe]6s
1
Francium Fr 87 [Rn]7s
1
Common oxidation state
All these elements are highly electropositive in nature. They readily lose their valence
electron to give monovalent cations (M
+
). Alkali metals have only one oxidation state which is +1.
Atomic and ionic radii
Being the first element of each period, alkali metals have the largest atomic and
ionic radii in their respective periods. On moving down the group, there is an increase in
the number of shells and, therefore, atomic and ionic radii increase. The monovalent ions
(M
+
) are smaller than the respective parent atoms as expected.
Table 5.3 Physical properties of alkali metals
Physical property
Li Na K Rb
Cs
Atomic radius / Metallic radius (Å) 1.52 1.86 2.27 2.48
2.65
Ionic radius (Å) 0.76 1.02 1.38 1.52
1.67
Melting point (
0
C) 181 98 63 39
28.5
Boiling point (
0
C) 1347 881 766 688
705
First ionization enthalpy (kJ mol
-1
) 520.2 495.8 418.8 403.0
375.7
Electronegativity (Paulings scale) 1.0 0.9 0.8 0.8
0.7
Density (g cm
-3
) 0.54 0.97 0.86 1.53
1.90
Standard potential E
Ѳ
for M
+
/M (V) -3.04 -2.71 -2.92 -2.93
-2.93
Hydration enthalpy (kJ mol
-1
) -506 -406 -330 -310
-276
128
Ionisation enthalpy
Alkali metals have the lowest
ionisation enthalpy compared to other
elements present in the respective period.
As we go down the group, the ionisation
enthalpy decreases due to the increase
in atomic size. In addition, the number
of inner shells also increases, which in
turn increases the magnitude of screening
effect and consequently, the ionisation
enthalpy decreases down the group.
The second ionisation enthalpies of
alkali metals are very high. The removal
of an electron from the alkali metals
gives monovalent cations having stable
electronic configurations similar to the
noble gas. Therefore, it becomes very
difficult to remove the second electron
from the stable configurations already
attained.
Hydration enthalpy
Lithium salts are more soluble
than the salts of other metals of group 1.
eg. LiClO
4
is up to 12 times more soluble
than NaClO
4
. KClO
4
, RbClO
4
and CsClO
4
have solubilities only 10
-3
times of that of
LiClO
4
. The high solubility of Li salts is
due to strong solvation of small size of Li
+
ion.
Li
+
Na
+
K
+
Rb
+
Cs
+
-519
-406
-322
-293
-264
enthalpy of hydration (kJ mol
-1
)
Figure 5.2 Hydration enthalphy of
alkali metals
Electronegativity:
Alkali metals have comparatively
smaller value of electronegativity than the
other elements in the respective period.
When they react with other elements,
they usually produce ionic compounds.
For example, they react with halogens to
form ionic halides.
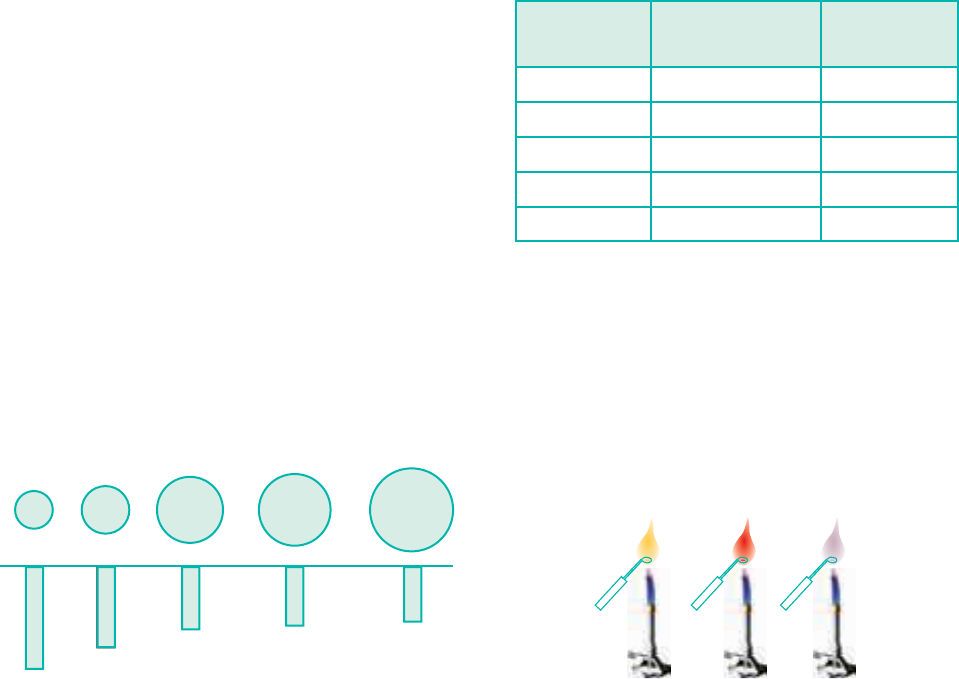
Flame colour and the spectra:
When the alkali metal salts
moistened with concentrated hydrochloric
acid are heated on a platinum wire in a
flame, they show characteristic coloured
flame as shown below.
Table 5.4 Flame colour and wavelength
Element Colour Wavelength
(nm)
Lithium Crimson red 670.8
Sodium Yellow 589.2
Potassium Lilac 766.5
Rubidium Reddish violet 780.0
Caesium Blue 455.5
The heat in the flame excites the
valence electron to a higher energy level.
When it drops back to its actual energy
level, the excess energy is emitted as light,
whose wavelength is in the visible region
as shown in the above table.
Sodium
Lithium
Potassium
Yellow Crimson Red Lilac
Figure 5.3 Flame colours of
alkali metal salts

129
5.2.2 Distinctive behavior of lithium
The distinctive behaviour of Li
+
ion is due to its exceptionally small size, high polarising
power, high hydration energy and non availability of d-orbitals.
Table 5.5 Comparison of properties of lithium with other elements of the group:
Lithium Other elements of the family
Hard, high melting and boiling point So and Lower melting and boiling point
Least reactive (For example it reacts with oxygen
to form normal oxide, forms peroxides with great
diculty and its higher oxides are unstable)
More reactive
Reacts with nitrogen to give Li
3
N No reaction
Reacts with bromine slowly React violently
Reacts directly with carbon to form ionic carbides.
For example 2Li + 2C --> Li
2
C
2
Do not react with carbon directly, but can react with
carbon compounds.
Na + C
2
H
2
--> Na
2
C
2
Compounds are sparingly soluble in water highly soluble in water.
Lithium nitrate decomposes to give an oxide decompose to give nitrites
Table 5.6 Similarities between lithium and Magnesium
S.No. Properties
1 Both lithium and magnesium are harder than other elements in the respective groups
2
Lithium and magnesium react slowly with water. eir oxides and hydroxides are much less
soluble and their hydroxides decompose on heating.
3 Both form a nitride, Li
3
N and Mg
3
N
2
, by direct combination with nitrogen
4 ey do not give any superoxides and form only oxides, Li
2
O and MgO
5
e carbonates of lithium and magnesium decompose upon heating to
form their respective oxides and CO
2
.
6 Lithium and magnesium do not form bicarbonates.
7
Both LiCl and MgCl
2
are soluble in ethanol and are deliquescent. ey crystallise from aqueous
solution as hydrates, LiCl·2H
2
O and MgCl
2
·8H
2
O
Diagonal Relationship:
Similarity between the first member of group 1 (Li) and the diagonally placed
second element of group 2 (Mg) is called diagonal relationship. It is due to similar
size (r Li
+
= 0.766 Å and Mg
2+
= 0.72 Å) and comparable electronegativity values
(Li = 1.0; Mg = 1.2).

130
5.2.3 Chemical properties of alkali
metals
Alkali metals exhibit high chemical
reactivity. The reactivity of alkali metals
increases from Li to Cs, since the ionisation
energy decreases down the group. All
alkali metals are highly reactive towards
the more electronegative elements such as
oxygen and halogens. Some characteristic
chemical properties of alkali metals are
described blow.
Reaction with oxygen
All the alkali metals on exposure
to air or oxygen burn vigorously, forming
oxides on their surface. Lithium forms only
monoxide, sodium forms the monoxide
and peroxide and the other elements form
monoxide, peroxide, and superoxides.
These oxides are basic in nature.
4 Li +O
2
2Li
2
O (simple oxide)
2 Na +O
2
Na
2
O
2
(peroxide)
M + O
2
MO
2
(M= K, Rb,Cs; MO
2
-superoxide)
Reaction with hydrogen
All alkali metals react with
hydrogen at about 673 K (lithium at
1073 K) to form the corresponding ionic
hydrides. Reactivity of alkali metals with
hydrogen decreases from Li to Cs.
2M + H
2
2 M
+
H
-
(M = Li, Na, K, Rb, Cs)
The ionic character of the hydrides
increases from Li to Cs and their stability
decreases. The hydrides behave as strong
reducing agents and their reducing nature
increases down the group.
Reaction with halogen
Alkali metals combine readily
with halogens to form ionic halides MX.
Reactivity of alkali metals with halogens
increases down the group because of
corresponding decrease in ionisation
enthalpy.
2M + X
2
2 MX
(M= Li, Na, K, Rb, Cs) (X= F, Cl, Br, I)
All metal halides are ionic crystals.
However Lithium iodide shows covalent
character, as it is the smallest cation that
exerts high polarising power on the iodide
anion. Additionally, the iodide ion being
the largest can be polarised to a greater
extent by Li
+
ion.
Reaction with liquid ammonia:
Alkali metals dissolve in liquid
ammonia to give deep blue solutions
that are conducting in nature. The
conductivity is similar to that of pure
metals (The specific conductivity of Hg is
10
4
Ω
-1
and for sodium in liquid ammonia
is 0.5 x 10
4
Ω
-1
). This happens because the
alkali metal atom readily loses its valence
electron in ammonia solution. Both the
cation and the electron are ammoniated to
give ammoniated cation and ammoniated
electron.
M + (x + y)NH
3
[M(NH
3
)
x
]
+
+ [e(NH
3
)
y
]
−
The blue colour of the solution is
due to the ammoniated electron which

131
absorbs energy in the visible region of
light and thus imparts blue colour to the
solution. The solutions are paramagnetic
and on standing slowly liberate hydrogen
resulting in the formation of an amide.
M
+
+ e
−
+ NH
3
MNH
2
+ ½H
2
In concentrated solution, the blue colour
changes to bronze colour and become
diamagnetic.
Reaction with water:
Alkali metals react with water to
give corresponding hydroxides with the
liberation of hydrogen.
2 Li + 2 H
2
O 2 LiOH+ H
2
They also react with alcohol, and
alkynes which contain active hydrogens.
2 Na + 2 C
2
H
5
OH 2 C
2
H
5
ONa + H
2
H-C C-H
Na
H-C C-Na
Na-C C-Na
Na
Reducing activity:
Alkali metals can lose their valence
electron readily hence they act as good
reducing agents.
M
(s)
M
+
(g)
+ e
–
Reaction with carbon:
Lithium directly reacts with carbon
to form the ionic compound, lithium
carbide. Other metals do not react with
carbon directly. However, when they are
treated with compounds like acetylene
they form acetelydes.
2 Li + 2C Li
2
C
2
5.2.4 Uses of alkali metals:
i. Lithium metal is used to make useful
alloys. For example with lead it is
used to make ‘white metal’ bearings
for motor engines, with aluminium
to make aircraft parts, and with
magnesium to make armour plates. It
is used in thermonuclear reactions.
ii. Lithium is also used to make
electrochemical cells.
iii. Lithium carbonate is used in medicines
iv. Sodium is used to make Na/Pb alloy
needed to make Pb(Et)
4
and Pb(Me)
4
.
These organolead compounds were
earlier used as anti-knock additives to
petrol, but nowadays lead-free petrol
in use.
v. Liquid sodium metal is used as
a coolant in fast breeder nuclear
reactors. Potassium has a vital role in
biological systems.
vi. Potassium chloride is used as a
fertilizer. Potassium hydroxide is used
in the manufacture of soft soap. It is
also used as an excellent absorbent of
carbon dioxide.
vii. Caesium is used in devising
photoelectric cells.
5.3 General characteristics of the
compounds of alkali metals
All the common compounds of the
alkali metals are generally ionic in nature.
General characteristics of some of their
compounds are discussed here.
132
Oxides and Hydroxides
On combustion in excess of air,
alkali metals forms normal oxides with
formula M
2
O. They react with water to
form corresponding hydroxides which are
basic in nature.
M
2
O + H
2
O 2 MOH
Alkali metals apart from lithium
form peroxides in addition to normal
oxides upon combustion with excess air.
These peroxides produce hydroxides and
H
2
O
2
upon reacting with water.
M
2
O
2
+2 H
2
O 2MOH+H
2
O
2
(M = Na, K, Rb, Cs)
Except lithium and sodium, all
the other alkali metals form superoxides
also. These superoxides also gives basic
hydroxides upon treatment with water.
2 MO
2
+ 2 H
2
O 2 MOH + H
2
O
2
+ O
2
(M = K, Rb, Cs)
Under appropriate conditions pure
compounds M
2
O, M
2
O
2
or MO
2
may be
prepared.
Properties of oxides and hydroxides:
The oxides and the peroxides are
colourless when pure, but the superoxides
are yellow or orange in colour. The
peroxides are diamagnetic while the
superoxides are paramagnetic. Sodium
peroxide is widely used as an oxidising
agent. The hydroxides which are obtained
by the reaction of the oxides with water
are all white crystalline solids. The alkali
metal hydroxides are strong bases. They
dissolve in water with evolution of heat on
account of intense hydration.
Halides:
The alkali metal halides, MX,
(X=F, Cl, Br, I) are colourless crystalline
solids with high melting points. They
can be prepared by the reaction of the
appropriate oxide, hydroxide or carbonate
with aqueous hydrohalic acid (HX). As the
electropositive character of alkali metal
increases from Li to Cs, the ease with
which the metals form halides increases
from Li to Cs. All halides are ionic in
nature except LiBr and LiI. Except LiF,
all other halides are soluble in water. The
low solubility of LiF in water is due to its
high lattice enthalpy (small size of Li
+
and
F
-
). Due to the presence of covalent nature
both LiBr and LiI are soluble in organic
solvents.
Salts of oxo-acids
Alkali metals form salts with all the
oxo-acids. Most of these salts are soluble
in water and are thermally stable. As the
electropositive character increases down
the group, the stability of the carbonates
and bicarbonates increases. This is due to
the decrease in polarising power of alkali
metal cations. The carbonates (M
2
CO
3
)of
alkali metals are remarkably stable up
to 1273 K, above which they first melt
and then eventually decompose to form
oxides. However, Li
2
CO
3
is considerably
less stable and decomposes readily.
Li
2
CO
3
Δ
Li
2
O+ CO
2
This is presumably due to large size
difference between Li
+
and CO
2
-3
which
133
makes the crystal lattice unstable. Being
strongly basic, alkali metals except lithium
form solid bicarbonates. No other metal
forms solid bicarbonates.
M
2
CO
3
+ CO
2
+ H
2
O 2 MHCO
3
(M = Na, K, Rb, Cs)
All the carbonates and bicarbonates
are soluble in water and their solubilities
increase rapidly on descending the group.
This is due to the reason that lattice
energies decrease more rapidly than their
hydration energies on moving down the
group.
5.3.1 Important compounds of alkali
metals:
Sodium Carbonate Na
2
CO
3
.10H
2
O (Washing
soda):
Sodium carbonate is one of the
important inorganic compounds used in
industries. It is prepared by Solvay process.
In this process, ammonia is converted
into ammonium carbonate which then
converted to ammonium bicarbonate by
passing excess carbon dioxide in a sodium
chloride solution saturated with ammonia.
The ammonium bicarbonate thus formed
reacts with the sodium chloride to give
sodium bicarbonate and ammonium
chloride. As sodium bicarbonate has poor
solubility, it gets precipitated. The sodium
bicarbonate is isolated and is heated to
give sodium carbonate. The equations
involved in this process are,
2NH
3
+ H
2
O + CO
2
(NH
4
)
2
CO
3
(NH
4
)
2
CO
3
+ H
2
O + CO
2
2 NH
4
HCO
3
2 NH
4
HCO
3
+ NaCl
NH
4
Cl + NaHCO
3
2 NaHCO
3
Na
2
CO
3
+ CO
2
+ H
2
O
The ammonia used in this process
can be recovered by treating the resultant
ammonium chloride solution with calcium
hydroxide. Calcium chloride is formed as
a by-product.
Properties:
Sodium carbonate, commonly
known as washing soda, crystallises as
decahydrate which is white in colour. It
is soluble in water and forms an alkaline
solution. Upon heating, it looses the water
of crystallisation to form monohydrate.
Above 373 K, the monohydrate becomes
completely anhydrous and changes to a
white powder called soda ash.
Na
2
CO
3
·10H
2
O Na
2
CO
3
·H
2
O + 9H
2
O
Na
2
CO
3
·H
2
O Na
2
CO
3
+ H
2
O
Uses:
i. Sodium carbonate known as washing
soda is used heavily for laundering
ii. It is an important laboratory reagent
used in the qualitative analysis and in
volumetric analysis.
iii. It is also used in water treatment to
convert the hard water to soft water
iv. It is used in the manufacturing of
glass, paper, paint etc...
Sodium chloride NaCl (Cooking salt or
Table salt):
Sodium chloride is isolated by
evaporation from sea water which contains
134
2.7 to 2.9% by mass. Approximately 50
lakh tons of salt are produced annually in
India by solar evaporation. Crude sodium
chloride can be obtained by crystallisation
of brine solution which contains sodium
sulphate, calcium sulphate, calcium
chloride and magnesium chloride as
impurities. Pure sodium chloride can
be obtained from crude salt as follows.
Firstly removal of insoluble impurities
by filtration from the crude salt solution
with minimum amount of water. Sodium
chloride can be crystallised by passing
HCl gas into this solution. Calcium and
magnesium chloride, being more soluble
than sodium chloride, remain in solution.
Sodium chloride melts at 1081K. It
has a solubility of 36.0 g in 100 g of water
at 273 K. The solubility does not increase
appreciably with increase in temperature.
Uses :
(i) It is used as a common salt or table
salt for domestic purpose.
(ii) It is used for the preparation of many
inorganic compounds such as NaOH
and Na
2
CO
3
Sodium hydroxide:
Sodium hydroxide is prepared
commercially by the electrolysis of brine
solution in Castner-Kellner cell using a
mercury cathode and a carbon anode.
Sodium metal is discharged at the cathode
and combines with mercury to form
sodium amalgam. Chlorine gas is evolved
at the anode. The sodium amalgam thus
obtained is treated with water to give
sodium hydroxide.
At cathode : Na
+
+ e
–
Na(amalgam)
At anode : Cl
–
½ Cl
2
↑+ e
–
2Na(amalgam)+2H
2
O2NaOH+2Hg+H
2
↑
Sodium hydroxide is a white,
translucent and deliquescent solid, that
dissolves in water to give a strong alkaline
solution. It melts at 591 K. The sodium
hydroxide solution at the surface reacts
with the CO
2
in the atmosphere to form
Na
2
CO
3
Uses:
• Sodium hydroxide is used as a
laboratory reagent
• It is also used in the purification of
bauxite and petroleum refining
• It is used in the textile industries for
mercerising cotton fabrics
• It is used in the manufacture of soap,
paper, artificial silk and a number of
chemicals
Sodium bicarbonate NaHCO
3
(Backing
soda):
Sodium hydrogen carbonate or
sodium bicarbonate is used in backing
cakes pastries etc. It is called so because
it decomposes on heating to generate
bubbles of carbon dioxide, leaving holes
in cakes or pastries and making them light
and fluffy. This compound is prepared by
saturating a solution of sodium carbonate
with carbon dioxide. The white crystalline
powder of sodium bicarbonate, being less
soluble, precipitated out.

135
Uses:
• Primarily used as an ingredient in
backing.
• Sodium hydrogen carbonate is a mild
antiseptic for skin infections.
• It is also used in fire extinguishers.
5.4 Biological importance of sodium
and potassium
Monovalent sodium and potassium
ions are found in large proportions in
biological fluids. These ions perform
important biological functions such as
maintenance of ion balance and nerve
impulse conduction. A typical 70 kg man
contains about 90 g of sodium and 170 g
of potassium compared with only 5 g of
iron and 0.06 g of copper.
Sodium ions are found primarily
on the outside of cells, being located
in blood plasma and in the interstitial
fluid which surrounds the cells. These
ions participate in the transmission of
nerve signals, in regulating the flow of
water across cell membranes and in the
transport of sugars and amino acids into
cells. Sodium and potassium, although so
similar chemically, differ quantitatively in
their ability to penetrate cell membranes,
in their transport mechanisms and in
their efficiency to activate enzymes. Thus,
potassium ions are the most abundant
cations within cell fluids, where they
activate many enzymes, participate in
the oxidation of glucose to produce ATP
and, with sodium, are responsible for the
transmission of nerve signals.
Sodium–potassium pump play an
important role in transmitting nerve signals.
Figure 5.4 Sodium–potassium pump
5.5 Alkaline earth metals
Group 2 in the modern periodic
table contains the elements beryllium,
magnesium, calcium, strontium, barium
and radium. These elements with the
exception of beryllium are commonly
known as the alkaline earth metals because
their oxides and hydroxides are alkaline in
nature and these metal oxides are found in
the earth’s crust.
Beryllium Magnesium
Calcium Strontium
Barium Radium
Figure 5.5 Alkaline earth metals

136
Table 5.7 Abundance of important alkaline
earth metals and their sources
Element
Abundance in Earth
crust by weight (ppm)
Mineral source
Be 2.0 beryl Be
3
Al
2
Si
6
O
18
Mg 27640
Carnallite (KCl.MgCl
2.
6H
2
O)
Dolomite MgCO
3
CaCO
3
Ca 1.84 Fluorapatite Ca
5
(PO
4
)
3
F
Sr 384 Celestite SrSO
4
Ba 390 barytes BaSO
4
5.5.1 General characteristics of alkaline
earth metals
Physical state
Beryllium is rare and radium is
the rarest of all comprising only 10 % of
igneous rocks. Magnesium and calcium
are very common in the earth’s crust,
with calcium the fifth-most-abundant
element, and magnesium the eighth.
Magnesium and calcium are found in
many rocks and minerals: magnesium
in carnallite, magnesite, dolomite and
calcium in chalk, limestone, gypsum.
Most strontium is found in the minerals
celestite and strontianite.Barium is slightly
less common, much of it in the mineral
barite. Radium, being a decay product of
uranium, is found in all uranium-bearing
ores.
FIREWORK
Many
alkaline
and
alkaline earth metals
are used in creating
colours, such as
strontium and barium, are the colourful
stars of a fireworks show. Combined
with the element chlorine, barium
sends up a green spark, calcium gives
orange and lithium gives medium red.
Strontium carbonate gives a bright
red colour Nitrates of sodium gives
orange, potassium and rubidium gives
violet colour and caesium gives indigo
colour. The burning "excites" the
electrons, pushing them into higher
than normal energy level; they release
their extra energy as a colourful burst
of light
The blue fireworks are the
hardest to make, since the compound
copper chloride breaks down in a
hot flame. In recent years, fireworks
experts have used magnalium- a
mixture of the alkaline earth metal
magnesium and aluminium - to boost
all firework colours. Magnalium has
made the blues brighter, but pyro
technicians are still searching for a
blue as brilliant as the red, green and
yellow colours.
Electronic configuration
These elements have two electrons
in the valence shell of their atoms, preceded
by the noble gas configuration. Their
general electronic configuration is written

137
as [Noble gas]ns
2
where ‘n’ represents the
valence shell.
Table 5.8 Electronic configuration of
alkaline earth metals
Element
Atomic
No.
Electronic
conguration
Be 4 [He]2s
2
Mg 12 [Ne]3s
2
Ca 20 [Ar]4s
2
Sr 38 [Kr]5s
2
Ba
56 [Xe]6s
2
Ra 88 [Rn]7s
2
Atomic and ionic radii
The atomic and ionic radii of
alkaline earth metals are smaller than
the corresponding members of thealkali
metals. This is due to the fact the Group 2
elements having a higher nuclear charge
that allows electrons to be attracted more
strongly towards the nucleus. On moving
down the group, the radii increases due
to gradual increase in the number of the
shells and the screening effect.
Common oxidation state
The group 2 elements have two
electrons in their valence shell and by
losing these electrons, they acquire the
stable noble gas configuration. So these
elements exhibit +2 oxidation state in
their compounds.
Ionisation enthalpy
Due to a fairly large size of the
atoms, alkaline earth metals have low
ionisation enthalpies when compared to
‘p’ block elements. Down the group the
ionisation enthalpy decreases as atomic
size increases. This is due to the addition
of new shells as well as increase in the
magnitude of the screening effect of inner
shell electrons. Members of group 2 have
higher ionization enthalpy values than
group 1 because of their smaller size, with
electrons being more attracted towards
the nucleus of the atoms. Correspondingly
they are less electropositive than alkali
metals.
Table 5.9 Physical properties of alkaline earth metals
Physical property Be Mg Ca Sr Ba
Atomic radius -non bonded (Å) 1.12 1.60 1.97 2.15 2.22
Ionic radius (Å) 0.27(0.31) 0.72 1.00 1.18 1.35
First ionization energy (kJ mol
-1
) 899.5 737.8 589.8 549.5 502.9
Second ionization energy (kJ mol
-1
) 1757.1 1450.7 1145.5 1064.2 965.2
Hydration enthalpy (kJ mol
-1
) – 2494 – 1921 –1577 – 1443 – 1305
Melting Point (
0
C) 1287 651 851 789 729
Boiling Point (
0
C) 2472 1090 1494 1382 1805
Density (g cm
-3
) 1.84 1.74 1.55 2.63 3.59
Standard Potential E
Ѳ
for M
+
/M (V) -1.97 -2.36 -2.84 -2.89 -2.92
Electronegativity (Paulings scale) 1.6 1.2 1.0 1.0 0.9

138
Ionisation Energy
Atomic number
Alkaline earth metal
899
738
590
550
503
Ra
Ba
Sr
Ca
Mg
Figure 5.6 Variation of ionisation
energy - Alkaline earth metals.
Although IE
1
values of alkaline
earth metals are higher than that of alkali
metals, the IE
2
values of alkaline earth
metals are much smaller than those of
alkali metals. This occurs because in
alkali metals the second electron is to be
removed from a cation, which has already
acquired a noble gas configuration. In
the case of alkaline earth metals, the
second electron is to be removed from a
monovalent cation, which still has one
electron in the outermost shell. Thus, the
second electron can be removed more
easily in the case of group 2 elements than
in group 1 elements.
Hydration Enthalpies
Compounds of alkaline earth metals
are more extensively hydrated than those
of alkali metals, because the hydration
enthalpies of alkaline earth metal ions
are larger than those of alkali metal ions.
Like alkali metal ions, the hydration
enthalpies of alkaline earth metal ions
also decrease with increase in ionic size
down the group.
Be > Mg > Ca > Sr > Ba
e.g., MgCl
2
and CaCl
2
exist as MgCl
2
.6H
2
O
and CaCl
2
· 6H
2
O while NaCl and KCl do
not form such hydrates.
Electronegativity
In alkaline earth metals the
electronegativity values decrease as we
go down the group as seen in the alkali
metals.
Flame colour and the spectra:
When the alkaline earth metal salts
moistened with concentrated hydrochloric
acid are heated on a platinum wire in a
flame, they show characteristic coloured
flame as shown below.
Table 5.10 Flame Colour and
wavelength
Element Colour Wavelength
(nm)
Calcium Brick - Red 622
Strontium Crimson 689
Barium Apple Green 554
The heat in the flame excites the
valence electron to a higher energy level.
when it drops back to its actual energy
level, the excess energy is emitted as light,
whose wavelength is in the visible region
as shown in the above table.

139
Calcium
Strontium
Barium
Brick - Red Crimson Red Apple Green
Figure 5.7: Flame colours of alkaline earth metal salts
5.5.2 Distinctive behavior of beryllium
Reason for the anomalous
behaviour of beryllium
Its small size and high
polarising power
Relatively high electronegativity
and ionisation enthalpy as
compared to other members
Absence of vacant d-orbitals in
its valence shell
Figure 5.8 Distinctive behaviour of beryllium
The anomalous properties of beryllium is mainly due to its small size, high
electronegativity, high ionisation energy and high polarising power compared to the other
elements in the block. The anomalous properties of beryllium compared to other elements
of the group are mentioned in Table 5.11
Table 5.11 Comparison of Properties of Beryllium with other elements of the group
Beryllium Other elements of the family
Forms covalent compounds form ionic compounds
High melting and boiling point Low melting and boiling point
Does not react with water even at elevated
temperature
React with water
Does not combine directly with hydrogen Combine directly with hydrogen
Does not combine directly with halogens.
Halides are covalent.
Combine directly with halogens
Halides are electrovalent.

140
Hydroxide and oxides of beryllium are
amphoteric in nature
Basic in nature.
It is not readily attacked by acids because of
the presence of an oxide lm
Readily attacked by acids
Beryllium carbide evolves methane with
water.
evolve acetylene with water.
Salts of Be are extensively hydrolysed Hydrolysed
Diagonal Relationship:
As observed in alkali metals, beryllium (the first member of group 2) shows a
diagonal relationship with aluminium. In this case, the size of these ions (r
Be
2+
= 0.45 Å
and r
Al
3+
= 0.54 Å) is not as close. However, their charge per unit area is closer (Be
2+
= 2.36
and Al
3+
= 2.50). They also have same electronegativity values (Be = 1.5; Al = 1.5).
Table 5.12 Similarities between Beryllium and Aluminium
S.No. Properties
1
Beryllium chloride forms a dimeric structure like aluminium chloride with
chloride bridges. Beryllium chloride also forms polymeric chain structure
in addition to dimer. Both are soluble in organic solvents and are strong
Lewis acids.
2
Beryllium hydroxide dissolves in excess of alkali and gives beryllate ion and
[Be(OH)
4
]
2–
and hydrogen as aluminium hydroxide which gives aluminate
ion, [Al(OH)
4
]
–
.
3
Beryllium and aluminum ions have strong tendency to form complexes,
BeF
4
2–
, AlF
6
3–
.
4
Both beryllium and aluminium hydroxides are amphoteric in nature.
5
Carbides of beryllium (Be
2
C) like aluminum carbide (Al
4
C
3
) give methane
on hydrolysis.
6
Both beryllium and aluminium are rendered passive by nitric acid.
5.5.3 Chemical properties of alkaline earth metals
The alkaline earth metals are less reactive than the alkali metals. The reactivity of
these elements increases on going down the group.
Reactivity towards the halogens:
All the alkaline earth metals combine with halogen at elevated temperatures to
form their halides.
M + X
2
MX
2

141
(M= Be, Mg, Ca, Sr, Ba, Ra ,
X = F, Cl, Br, l )
Thermal decomposition of
(NH
4
)
2
BeF
4
is the best route for the
preparation of BeF
2
. BeCl
2
is conveniently
made from the oxide.
BeO + C + Cl
2
BeCl
2
+ CO
600 − 800K
Reactivity towards hydrogen:
All the elements except beryllium,
combine with hydrogen on heating to form
their hydrides with general formula MH
2
.
BeH
2
can be prepared by the reaction of
BeCl
2
with LiAlH
4
.
2BeCl
2
+ LiAlH
4
2BeH
2
+ LiCl + AlCl
3
5.5.4 Uses of alkaline earth metals
Uses of beryllium
1. Because of its low atomic number and
very low absorption for X-rays, it is
used as radiation windows for X-ray
tubes and X-ray detectors.
2. The sample holder in X-ray emission
studies usually made of beryllium
3. Since beryllium is transparent to
energetic particles it is used to build
the ‘beam pipe’ in accelerators.
4. Because of its low density and
diamagnetic nature, it is used in
various detectors.
Uses of magnesium
1. Removal of sulphur from iron and steel
2. Refining of titanium in the “Kroll”
process.
3. Used as photoengrave plates in printing
industry.
4. Magnesium alloys are used in aeroplane
and missile construction.
5. Mg ribbon is used in synthesis of
Grignard reagent in organic synthesis.
6. It alloys with aluminium to improve its
mechanical, fabrication and welding
property.
7. As a desiccant .
8. As sacrificial anode in controlling
galvanic corrosion.
Uses of calcium
1. As a reducing agent in the metallurgy
of uranium, zirconium and thorium.
2. As a deoxidiser, desulphuriser or
decarboniser for various ferrous and
non-ferrous alloys.
3. In making cement and mortar to be
used in construction.
4. As a getter in vacuum tubes.
5. In dehydrating oils
6. In fertilisers, concrete and plaster of
paris.
Uses of strontium
1.
90
Sr is used in cancer therapy.

142
2.
87
Sr /
86
Sr ratios are commonly used
in marine investigations as well as in
teeth, tracking animal migrations or in
criminal forensics.
3. Dating of rocks.
4. As a radioactive tracer in determining
the source of ancient archaeological
materials such as timbers and coins.
Uses of Barium
1. Used in metallurgy, its compounds
are used in pyrotechnics, petroleum
mining and radiology.
2. Deoxidiser in copper refining.
3. Its alloys with nickel readily emits
electrons hence used in electron tubes
and in spark plug electrodes.
4. As a scavenger to remove last traces of
oxygen and other gases in television
and other electronic tubes.
5. An isotope of barium
133
Ba, used as a
source in the calibration of gamma ray
detectors in nuclear chemistry.
Uses of Radium
Used in self-luminous paints for
watches, nuclear panels, aircraft switches,
clocks and instrument dials.
5.6. General characteristics of the
compounds of the alkaline earth metals
The dipositive oxidation state (M
2+
)
is the predominant valence of group 2
elements. The alkaline earth metals form
compounds which are predominantly
ionic. However, they are less ionic than
the corresponding compounds of alkali
metals. This is due to increased nuclear
charge and smaller size. The general
characteristics of some of the compounds
of alkaline earth metals are described
below.
(a) Oxides
Generally alkaline earth metals
form monoxides and peroxides.
Monoxides
Monoxides are obtained by heating
the metals in oxygen. BeO and MgO are
almost insoluble in water. On the other
hand, oxides of other elements form
hydroxides. BeO is amphoteric; MgO is
weakly basic while CaO, SrO and BaO are
strongly basic.
BeO oxide is covalent due to the
small size of Be
2+
ion,while other oxides
are ionic in nature.
Peroxides
Except beryllium, all the remaining
metals form peroxides. It is prepared by
heating monoxides with oxygen at high
temperature.
2 BaO +O
2
2 BaO
2
b)Hydroxides:
All the oxides except BeO are basic
in nature and react with water to form
sparingly soluble hydroxides.
MO + H
2
O →M(OH)
2

143
The solubility, thermal stability
and the basic character of the hydroxides
increase down the group. The alkaline
earth metal hydroxides are, however,
less basic and less stable than alkali
metal hydroxides. Beryllium hydroxide
is amphoteric in nature as it reacts with
both acid and alkali.
Be(OH)
2
+ 2 NaOH → Na
2
BeO
2
+2H
2
O
Be(OH)
2
+ 2HCl → BeCl
2
+2H
2
O
c) Halides:
Alkaline earth metals form halides
with general formula MX
2
. They can be
prepared by heating metals with halogens
on heating.
M +X
2
MX
2
Beryllium halides are covalent on
account of smaller size of Be
+2
. Beryllium
halides are hygroscopic, fume in moist air
and soluble in organic solvents. Beryllium
chloride has a chain structure in the solid
state as shown in figure 5.9 (structure-a).
In the vapour phase BeCl
2
t e n d s t o f o r m
a chloro-bridged dimer (structure-c)
which dissociates into the linear monomer
at high temperatures of the order of 1200 K.
(structure-b).
Except beryllium halides, all the
other halides of alkaline earth metals are
ionic in nature. Chloride and fluorides of
the other metals are ionic solids. These are
good conductors of electricity in fused state
and in aqueous solutions. The tendency to
form halide hydrates gradually decreases
(for example, MgCl
2.
.8H
2
O, CaCl
2
.6H
2
O,
SrCl
2
.6H
2
O and BaCl
2
.2H
2
O) down the
group.
Be
Be Be
Be
Be Be
Be Be
Be
Figure 5.9 Structure of beryllium
chloride
Salts of oxo acids
The alkaline earth metals form salts of
oxo acids. Some of these are given below:
Carbonates:
All the carbonates decompose on
heating to give carbon dioxide and the
oxide.
MCO
3
MO + CO
2
Δ
• The solubility of carbonates in water
decreases down the group.
• The thermal stability increases down
the group with increasing cationic size.
Table 5.13 Decomposition temperature
of alkaline metal carbonates and
sulphates
Element
Decomposition
temp for
carbonates
(in
o
C)
Decomposition
temp for
sulphates
(in
o
C)
Be 25 500
Mg 540 895
Ca 900 1149
Sr 1290 1374
Ba 1360 -

144
Sulphates:
The sulphates of the alkaline earth
metals are all white solids and stable to
heat. BeSO
4
, and MgSO
4
are readily soluble
in water; the solubility decreases from
CaSO
4
to BaSO
4
. The greater hydration
enthalpies of Be
2+
and Mg
2+
ions overcome
the lattice enthalpy factor and therefore
their sulphates are soluble in water.
Nitrates:
The nitrates are made by dissolution
of the carbonates in dilute nitric acid.
Magnesium nitrate crystallises with six
molecules of water, whereas barium
nitrate crystallises as the anhydrous salt.
This again shows a decreasing tendency
to form hydrates with increasing size. All
of them decompose on heating to give the
oxide.
5.6.1 Important compounds of calcium
Quick lime, CaO
Preparation
It is produced on a commercial
scale by heating limestone in a lime kiln
in the temperature range 1070-1270K.
CaCO
3
֖ CaO + CO
2
The reaction being reversible,
carbon dioxide is removed as soon as
it is produced to enable the reaction to
proceed to completion.
Properties
Calcium oxide is a white amorphous solid.
It has a melting point of 2870 K.
It absorbs moisture and carbon dioxide on
exposure to atmosphere.
CaO + H
2
O Ca(OH)
2
CaO + CO
2
CaCO
3
The addition of limited amount
of water breaks the lump of lime. This
process is called slaking of lime and the
product is slaked lime.
CaO + H
2
O Ca(OH)
2
Quick lime mixed with soda gives
solid soda lime. It combines with acidic
oxides such as SiO
2
and P
4
O
10
to form
CaSiO
3
and Ca
3
(PO
4
)
2
, respectively.
limestone
and coal
mixture in
waste gas
(CO
2
)
fuel
fuel
air in
lime out
Figure 5.10 Preparation of Quick Lime

145
CaO + SiO
2
CaSiO
3
6CaO + P
4
O
10
2Ca
3
(PO
4
)
2
Uses
Calcium oxide is used
(i) to manufacture cement, mortar and
glass.
(ii) in the manufacture of sodium
carbonate and slaked lime.
(iii) in the purification of sugar.
(iv) as a drying agent.
5.6.2 Calcium hydroxide
Preparation
Calcium hydroxide is prepared by
adding water to quick lime, CaO.
Properties
It is a white powder. It is sparingly
soluble in water. The aqueous solution is
known as lime water and a suspension of
slaked lime in water is known as milk of
lime.
When carbon dioxide is passed
through lime water, it turns milky due to
the formation of calcium carbonate.
Ca(OH)
2
+ CO
2
CaCO
3
+ H
2
O
On passing excess of carbon
dioxide, the precipitate dissolves to form
calcium hydrogen carbonate.
CaCO
3
+ CO
2
+ H
2
O Ca(HCO
3
)
2
Milk of lime reacts with chlorine
to form hypochlorite, a constituent of
bleaching powder.
2Ca (OH)
2
+ 2Cl
2
CaCl
2
+ Ca(OCl)
2
+ 2H
2
O
Uses:
Calcium hydroxide is used
(i) in the preparation of mortar, a
building material.
ii) in white wash due to its disinfectant
nature.
(iii) in glass making, in tanning industry,
in the preparation of bleaching
powder and for the purification of
sugar.
5.6.3 Gypsum (CaSO
4
.2H
2
O)
Gypsum beds were formed due
to the evaporation of water from the
massive prehistoric sea basins. When
water evaporates, the minerals present in
it become concentrated, and crystallise.
Figure 5.11 A Gypsum Quarry
Properties of Gypsum
• Gypsum is a soft mineral, which is
moderately soluble in water. The
solubility of this mineral in water is

146
affected by temperature. Unlike other
salts, gypsum becomes less soluble in
water as the temperature increases.
This is known as retrograde solubility,
which is a distinguishing characteristic
of gypsum.
• Gypsum is usually white, colorless, or
gray in color. But sometimes, it can
also be found in the shades of pink,
yellow, brown, and light green, mainly
due to the presence of impurities.
• Gypsum crystals are sometimes found
to occur in a form that resembles
the petals of a flower. This type of
formation is referred to as ‘desert rose’,
as they mostly occur in arid areas or
desert terrains.
• Gypsum is known to have low thermal
conductivity, which is the reason
why it is used in making drywalls or
wallboards. Gypsum is also known as
a natural insulator.
Figure 5.12 -The Alabaster Variety of
Gypsum
• Alabaster is a variety of gypsum, that is
highly valued as an ornamental stone. It has
been used by the sculptors for centuries.
Alabaster is granular and opaque.
• Gypsum has hardness between 1.5 to
2 on Moh’s Hardness Scale. Its specific
gravity is 2.3 to 2.4.
Uses of Gypsum
• The alabaster variety of gypsum was
used in ancient Egypt and Mesopotamia
by the sculptors. The ancient Egyptians
knew how to turn gypsum into plaster
of Paris about 5,000 years ago. Today,
gypsum has found a wide range of uses
and applications in human society,
some of which are enlisted below.
• Gypsum is used in making drywalls or
plaster boards. Plaster boards are used
as the finish for walls and ceilings, and
for partitions.
• Another important use of gypsum
is the production of plaster of Paris.
Gypsum is heated to about 300 degree
Fahrenheit to produce plaster of Paris,
which is also known as gypsum plaster.
It is mainly used as a sculpting material.
• Gypsum is used in making surgical
and orthopedic casts, such as surgical
splints and casting moulds.
• Gypsum plays an important role
in agriculture as a soil additive,
conditioner, and fertilizer. It helps
loosen up compact or clay soil, and
provides calcium and sulphur, which
are essential for the healthy growth
of a plant. It can also be used for
removing sodium from soils having
excess salinity.

147
• Gypsum is used in toothpastes, shampoos, and hair products, mainly due to its binding
and thickening properties.
• Gypsum is a component of Portland cement, where it acts as a hardening retarder to
control the speed at which concrete sets.
• To sum up, gypsum is one of the most abundant minerals that have endless uses and
applications. Mining of gypsum is simple and easy, as the mineral occurs in large
thick beds near the Earth’s surface. However, large-scale mining of gypsum involves
considerable damage to the environment. Gypsum can also be recycled, but not much
importance has been given to recycle this mineral due to its abundance.
Figure 5.13 -Uses of Gypsum
5.6.4 Plaster of paris
Calcium Sulphate (Plaster of Paris), CaSO
4
·½ H
2
O
It is a hemihydrate of calcium sulphate. It is obtained when gypsum, CaSO
4
·2H
2
O,
is heated to 393 K.
2CaSO
4
.2H
2
O(s) 2CaSO
4
.H
2
O+ 3H
2
O
Above 393 K, no water of crystallisation is left and anhydrous calcium sulphate,
CaSO
4
is formed. This is known as ‘dead burnt plaster’.

148
It has a remarkable property of setting with water. On mixing with an adequate
quantity of water it forms a plastic mass that gets into a hard solid in 5 to 15 minutes.
Uses:
The largest use of Plaster of Paris is in the building industry as well as plasters. It is
used for immobilising the affected part of organ where there is a bone fracture or sprain.
It is also employed in dentistry, in ornamental work and for making casts of statues and
busts.
5.7 Biological importance of magnesium and calcium
Magnesium and calcium also plays a vital role in biological functions. A typical
adult human body contains about 25 g of magnesium and 1200 g of calcium. Magnesium
plays an important role in many biochemical reactions catalysed by enzymes. It is the
co-factor of all enzymes that utilize ATP in phosphate transfer and energy release. It also
essential for DNA synthesis and is responsible for the stability and proper functioning
of DNA. It is also used for balancing electrolytes in our body. Deficiency of magnesium
results into convulsion and neuromuscular irritation.
Calcium is a major component of bones and teeth. It is also present in in blood
and its concentration is maintained by hormones (calcitonin and parathyroid hormone).
Deficiency of calcium in blood causes it to take longer time to clot. It is also important for
muscle contraction.
The main pigment that is responsible for photosynthesis, chlorophyll, contains
magnesium which plays an important role in photosynthesis.

149
SUMMARY
The elements belonging to groups 1
and 2 of the modern periodic table are
called s-block elements. They are called
so because the valence electron occupies
the s orbitals. The group 1 elements have
a general outer electronic configuration
ns
1
and are called alkali metals. The
group 2 elements have a general outer
electronic configuration ns
2
and these
are called alkaline earth metals as they
are found in earth’s crust and their oxides
and hydroxides are alkaline in nature.
Elements belonging to group 1 and 2 are
highly reactive and forms M
+
and M
2+
cations respectively. Their physical and
chemical properties of both groups show
a regular trend as we move down the
group. The atomic and ionic radii increase
as we move down the group while their
ionisation enthalpies decrease.
The first element in each of these
groups, lithium in Group 1 and beryllium
in Group 2 shows some difference in
behaviour with the elements in rest of their
groups and show similarities in properties
to the second member of the next group.
This behaviour is known as the ‘diagonal
relationship’ in the periodic table.
The alkali metals are soft and
silvery white in colour with low melting
points. They are highly reactive.
The compounds of alkali metals are
predominantly ionic. They form metal
hydrides and halides with hydrogen and
halogens respectively. Their oxides and
hydroxides are soluble in water forming
strong alkalies. Important compounds of
sodium include sodium carbonate, sodium
chloride, sodium hydroxide and sodium
hydrogen carbonate. Sodium hydroxide is
manufactured by Castner-Kellner process
and sodium carbonate by Solvay process.
The chemistry of alkaline earth
metals is similar to alkali metals.
However, we observe some differences
because of their reduced atomic and ionic
sizes and increased cationic charges.
Their oxides and hydroxides are less
basic than the alkali metal oxides and
hydroxides. They also form hydrides
and halides with hydrogen and halogens
respectively. Industrially important
compounds of calcium include calcium
oxide (lime), calcium hydroxide (slaked
lime), calcium sulphate hemihydrate
(Plaster of Paris), calcium carbonate
(limestone) and cement. Portland cement
is an important constructional material. It
is manufactured by heating a pulverised
mixture of limestone and clay in a rotary
kiln. The clinker thus obtained is mixed
with some gypsum (2-3%) to give a fine
powder of cement. All these substances
find variety of uses in different areas.
Monovalent sodium and potassium
ions and divalent magnesium and calcium
ions are found in large proportions in
biological fluids. These ions perform
important biological functions such as
maintenance of ion balance and nerve
impulse conduction.

150
EVALUATION
1. For alkali metals, which one of the following trends is incorrect ?
a) Hydration energy : Li > Na > K > Rb
b) Ionisation energy : Li > Na > K > Rb
c) Density : Li < Na < K < Rb
d) Atomic size : Li < Na < K < Rb
2. Which of the following statements is incorrect ?
a) Li
+
has minimum degree of hydration among alkali metal cations.
b) The oxidation state of K in KO
2
is +1
c) Sodium is used to make Na / Pb alloy
d) MgSO
4
is readily soluble in water
3. Which of the following compounds will not evolve H
2
gas on reaction with alkali
metals ?
a) ethanoic acid b) ethanol
c) phenol d) none of these
4. Which of the following has the highest tendency to give the reaction
M
+
(g)
Aqueous
Medium
M
+
(aq)
a) Na b) Li c) Rb d) K
5. sodium is stored in
a) alcohol b) water c) kerosene d) none of these
6. RbO
2
is
a) superoxide and paramagnetic b) peroxide and diamagnetic
c) superoxide and diamagnetic d) peroxide and paramagnetic
151
7. Find the wrong statement
a) sodium metal is used in organic qualitative analysis
b) sodium carbonate is soluble in water and it is used in inorganic qualitative
analysis
c) potassium carbonate can be prepared by solvay process
d) potassium bicarbonate is acidic salt
8. Lithium shows diagonal relationship with
a) sodium b) magnesium c) calcium d) aluminium
9. Incase of alkali metal halides, the ionic character increases in the order
a) MF < MCl < MBr < MI
b) MI < MBr < MCl < MF
c) MI < MBr < MF < MCl
d) none of these
10. In which process, fused sodium hydroxide is electrolysed for extraction of sodium ?
a) Castner's process b) Cyanide process
c) Down process d) All of these
11. The product obtained as a result of a reaction of nitrogen with CaC
2
is (NEET -
Phase I)
a) Ca(CN)
3
b) CaN
2
c) Ca(CN)
2
d) Ca
3
N
2
12. Which of the following has highest hydration energy
a) MgCl
2
b) CaCl
2
c) BaCl
2
d) SrCl
2
152
13. Match the flame colours of the alkali and alkaline earth metal salts in the bunsen
burner
(p) Sodium (1) Brick red
(q) Calcium (2) Yellow
(r) Barium (3) Violet
(s) Strontium (4) Apple green
(t) Cesium (5) Crimson red
(u) Potassium (6) Blue
a) p - 2, q - 1, r - 4, s - 5, t - 6, u - 3
b) p - 1, q - 2, r - 4, s - 5, t - 6, u - 3
c) p - 4, q - 1, r - 2, s - 3, t - 5, u - 6
d) p - 6, q - 5, r - 4, s - 3, t - 1, u - 2
14. Assertion : Generally alkali and alkaline earth metals form superoxides
Reason : There is a single bond between O and O in superoxides.
a) both assertion and reason are true and reason is the correct explanation of asser-
tion
b) both assertion and reason are true but reason is not the correct explanation of
assertion
c) assertion is true but reason is false
d) both assertion and reason are false
15. Assertion : BeSO
4
is soluble in water while BaSO
4
is not
Reason : Hydration energy decreases down the group from Be to Ba and
lattice energy remains almost constant.
a) both assertion and reason are true and reason is the correct explanation of asser-
tion
153
b) both assertion and reason are true but reason is not the correct explanation of
assertion
c) assertion is true but reason is false
d) both assertion and reason are false
16. Which is the correct sequence of solubility of carbonates of alkaline earth metals ?
a) BaCO
3
> SrCO
3
> CaCO
3
> MgCO
3
b) MgCO
3
> CaCO
3
> SrCO
3
> BaCO
3
c) CaCO
3
> BaCO
3
> SrCO
3
> MgCO
3
d) BaCO
3
> CaCO
3
> SrCO
3
> MgCO
3
17. In context with beryllium, which one of the following statements is incorrect ?
(NEET Phase - 2)
a) It is rendered passive by nitric acid
b) It forms Be
2
C
c) Its salts are rarely hydrolysed
d) Its hydride is electron deficient and polymeric
18. The suspension of slaked lime in water is known as (NEET Phase - II)
a) lime water b) quick lime
c) milk of lime d) aqueous solution of slaked lime
19. A colourless solid substance (A) on heating evolved CO
2
and also gave a white resi-
due, soluble in water. Residue also gave CO
2
when treated with dilute HCl.
a) Na
2
CO
3
b) NaHCO
3
c) CaCO
3
d) Ca(HCO
3
)
2
20. The compound (X) on heating gives a colourless gas and a residue that is dissolved
in water to obtain (B). Excess of CO
2
is bubbled through aqueous solution of B, C is
formed. Solid (C) on heating gives back X. (B) is
a) CaCO
3
b) Ca(OH)
2
c) Na
2
CO
3
d) NaHCO
3
21. Which of the following statement is false ? (NEET - Phase - I)
154
a) Ca
2+
ions are not important in maintaining the regular beating of the heart
b) Mg
2+
ions are important in the green parts of the plants
c) Mg
2+
ions form a complex with ATP
d) Ca
2+
ions are important in blood clotting
22. The name 'Blue John' is given to which of the following compounds ?
a) CaH
2
b) CaF
2
c) Ca
3
(PO
4
)
2
d) CaO
23. Formula of Gypsum is
a) CaSO
4
. 2H
2
O b) CaSO
4
. ½ H
2
O
c) 3 CaSO
4
. H
2
O d) 2CaSO
4
. 2H
2
O
24. When CaC
2
is heated in atmospheric nitrogen in an electric furnace the compound
formed is
a) Ca(CN)
2
b) CaNCN
c) CaC
2
N
2
d) CaNC
2
25. Among the following the least thermally stable is
(a) K
2
CO
3
b) Na
2
CO
3
(c) BaCo
3
d) Li
2
CO
3
26. Why sodium hydroxide is much more water soluble than chloride?
27. Explain what to meant by efflorescence;
28. Write the chemical equations for the reactions involved in solvay process of
preparation of sodium carbonate.
29. An alkali metal (x) forms a hydrated sulphate, X
2
SO
4
. 10H
2
O. Is the metal more
likely to be sodium (or) potassium.
30. Write balanced chemical equation for each of the following chemical reactions.
(i) Lithium metal with nitrogen gas
(ii) heating solid sodium bicarbonate
155
(iii) Rubidum with oxgen gas
(iv) solid potassium hydroxide with CO
2
(v) heating calcium carbonate
(vi) heating calcium with oxygen
31. Discuss briefly the similarities between beryllium and aluminium.
32. Give the systematic names for the following
(i) milk of magnesia (ii) lye (iii) lime (iv) Caustic potash
(v) washing soda (vi) soda ash
(v) trona
33. Substantiate Lithium fluoride has the lowest solubility among group one metal
fluorides.
34. Mention the uses of plaster of paris
35. Beryllium halides are Covalent whereas magnesium halides are ionic why?
36. Alkaline earth metal (A), belongs to 3rd period reacts with oxygen and nitrogen to
form compound (B) and (C) respectively. It undergo metal displacement reaction
with AgNO
3
solution to form compound (D).
37. Write balanced chemical equation for the following processes
(a) heating calcium in oxygen
(b) heating calcium carbonate
(c) evaporating a solution of calcium hydrogen carbonate
(d) heating calcium oxide with carbon
38. Explain the important common features of Group 2 elements.
39. Discus the similarities between beryllium and aluminium.
40. Why alkaline earth metals are harder than alkali metals.
41. How is plaster of paris prepared?
42. Give the uses of gypsum.
43. Describe briefly the biological importance of Calcium and magnesium.
44. Which would you expect to have a higher melting point, magnesium oxide or
magnesium fluoride? Explain your reasoning.
156
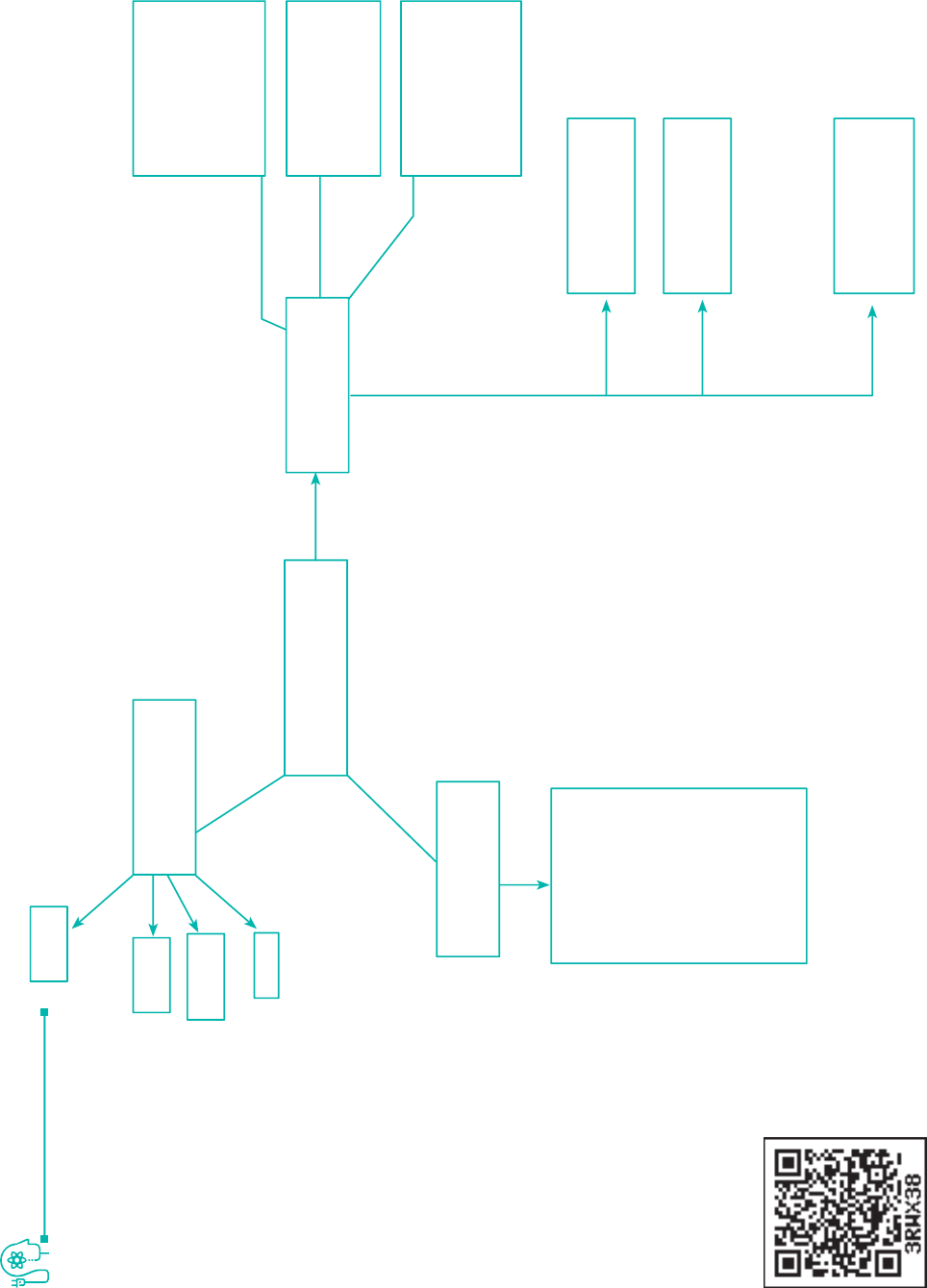
Important
Compounds
Chemical
Properties
Li
2
O
Na
2
O
2
Fluoride
MO
2
M = K, R, C
+O
2
+H
2
+X
2
+H
2
+C
CCM
(M =
Na, K,
Rb, Cs)
MH
(M= Li, K, Na,
Rh, Cs)
Mx
M= Li, Na, K, Cr,
Cs
X = F, Cl, Br, I
MoH (Li, Na, K,
Cr, Cs)
Li
MC CM
MNaK
RbCs
Physical
Properties
Common
Oxidation
Increases down
group
Atomic & Ionic
Radri
Decreases down
the group
IE, HE, EN
Alkali Metals
Lu, Na, K, Rb, Cs, Fr
NaOH
Na
2
CO
3
NaHCO
3
NaCl
CONCEPT MAP

157
Flame test of alkali and alkaline earth elements (Virtual Lab)
Step – 1
Open the Browser and type the URL given (or) Scan the QR Code. Just click the view button on the Flame test
panel. This will open a flame test window as shown in the figure.
Step – 2
Follow the instrutions to perform a virtual flame test.
1. Click on the wire loop (1).
2. Move the wire loop to the cleaning solution (2). Click on the cleaning solution. The wire must be cleaned
before each test to ensure there is no other salt on the loop.
3. Move the wire loop to the salt solution you want to test (3). Click on the salt solution.
4. Move the wire loop to the flame (4) and click on it.
5. You will see the characteristic colour change in the flame with respect to the metal ion
By using this virtual lab you
can perform the ame test of
di erent alkali and alkali earth
metals and see the colour of the
ame produced.
Please go to the URL
https://www.newpathonline.com/
free-curriculumresources/virtual_
lab/Flame_Test/9/12,13,14/1914
(or)
Scan the QR code on the right side
ICT Corner
